A recent study demonstrated the safety of cilostazol in patients with mild cognitive impairment (MCI), but did not demonstrate any efficacy in preventing disease progression. However, after administration of cilostazol, the blood concentrations of the albumin and β-amyloid complex (albumin-Aβ complex) increased in the treated patients compared with those receiving placebo.
This finding indicates that cilostazol may promote the clearance of β-amyloid—a waste product that accumulates in the brains of patients with dementia—into the blood. In previous studies, the researchers demonstrated the effect of cilostazol metabolites in improving cognitive function. Therefore, in the future, they plan to identify a group in which cilostazol is effective (cilostazol-responders), to enable investigation of its anti-dementia effects.
The work is published in the journal JAMA Network Open.
Currently, more than 5 million dementia patients are in Japan, and the MCI patient total is soon estimated to reach a similar number. A global initiative is presently underway to develop methods to halt the progression of this disease.
Previously, a group led by Masafumi Ihara, Director of the NCVC Department of Neurology, discovered through animal experiments that the antiplatelet drug cilostazol promoted the elimination of β-amyloid, which accumulates in the brains of many patients with dementia. However, whether cilostazol is effective for preventing dementia in humans remains unclear, emphasizing the need for verification.
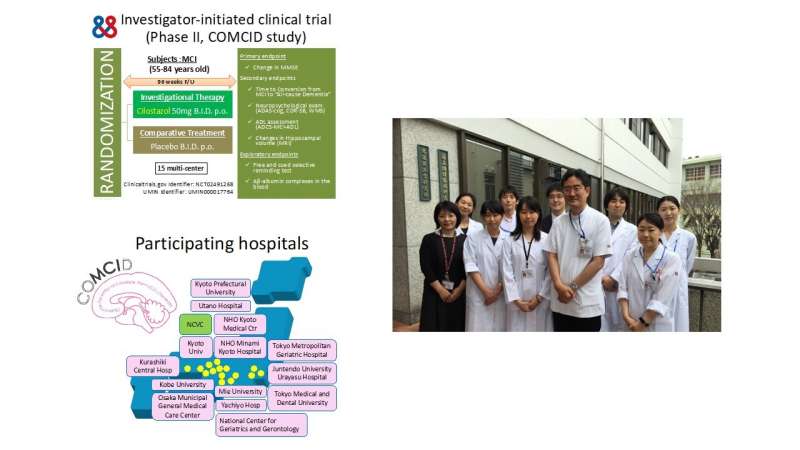
In May 2015, a research group led by Director Masafumi Ihara began conducting a nationwide, multicenter, investigator-initiated, double-blind, and randomized trial termed COMCID (cilostazol for the prevention of Conversion from MCI to Dementia). In the COMCID study, patients with MCI received either cilostazol or a placebo for 96 weeks.
This study demonstrated that cilostazol was safe when administered to patients with MCI; however, it did not prevent progression of MCI to dementia. Nevertheless, the blood concentrations of the albumin and β-amyloid complex (albumin-Aβ complex) in patients receiving cilostazol increased following treatment compared with those receiving placebos. This suggests that cilostazol may have promoted the clearance of β-amyloid from the brain into the blood, consistent with the results of past animal experiments.
This study did not demonstrate that cilostazol was beneficial for all patients with MCI. Consequently, in future research, the researchers plan to identify a group of patients in which cilostazol is effective (cilostazol-responders) to enable exploration of its anti-dementia effects in specific patients. However, this study did establish an important clinical trial-ready cohort for future research investigating cognitive dysfunction.
Based on clinical trials conducted in Japan, the COMCID cohort has great significance and enables further exploration of the efficacy and safety of anti-dementia drug candidates developed worldwide.
More information:
Satoshi Saito et al, Efficacy and Safety of Cilostazol in Mild Cognitive Impairment, JAMA Network Open (2023). DOI: 10.1001/jamanetworkopen.2023.44938
Journal information:
JAMA Network Open
Source: Read Full Article
