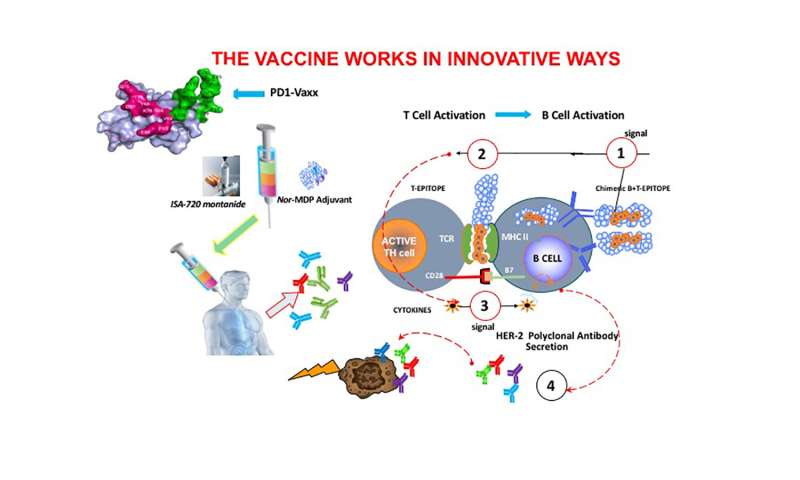
A study led by researchers at The Ohio State University Comprehensive Cancer Center—Arthur G. James Cancer Hospital and Richard J. Solove Research Institute (OSUCCC—James) described a potential therapeutic anticancer vaccine that frees suppressed cancer-killing immune cells, enabling them to attack and destroy a tumor.
Published in the journal Oncoimmunology, on October 1, 2020, the findings showed that the peptide called PD1-Vaxx, a first checkpoint inhibitor vaccine, was safe and effective in a colon cancer syngeneic animal model.
The vaccine produced polyclonal antibodies that inhibit the programmed cell death receptor, PD-1, on cancer cells. The vaccine mimics the action of the PD-1 inhibitor nivolumab (pronounced nih-VOL-yoo-mab, marketed as Opdivo), but it avoids triggering the innate and acquired resistance associated with that and related agents, the researchers say.
The study found that PD1-Vaxxwas effective in inhibiting tumor growth. It was even more effective when used in combination with a second therapeutic peptide vaccine, one that targets two sites on the HER-2 receptor on colon cancer cells. The combination treatment produced complete responses in nine of 10 animals. That vaccine, called B-Vaxx, was developed earlier by the same research team.
“Our study is important for two key reasons,” says first author and vaccine developer Pravin T. P. Kaumaya, Ph.D., a member of the OSUCCC—James Translational Therapeutics Research Program and professor of medicine at The Ohio State College of Medicine. “First, PD1-Vaxx activates both B- and T-cell functions to promote tumor clearance. Second, the treatment is targeted to block signaling pathways that are crucial for tumor growth and maintenance. By giving this vaccine in combination with an immunotherapy drug, we are essentially super-charging and specifically directing the immune system to target and kill cancer cells.”
Like the immune therapy drug nivolumab, PD1-Vaxx is an immune checkpoint inhibitor. Immune checkpoints are proteins that keep immune cells from attacking healthy body cells. PD-1 is a checkpoint protein on killer T cells. PD-L1 is another checkpoint protein that is on healthy cells and on many cancer cells. When PD-1 on T cells binds with PD-L1 on a body cell or a cancer cell, it suppresses the T cell, preventing it from killing the cell.
Nivolumab works by blocking PD-1 from binding with PD-L1, thereby allowing T cells to kill a patient’s cancer cells. But while nivolumab consists of anti-PD-1 monoclonal antibodies, which target a single location on the PD-1 protein, the experimental vaccine PD1-Vaxx triggers a range of antibodies—a polyclonal antibody response—that blocks multiple sites on PD-1 and could more effectively inhibit the protein.
For this study, Kaumaya and his colleagues used cell lines and animal models to evaluate four PD-1 B-cell peptide epitopes as vaccine candidates. Of these, the PD-1 epitope sequence 92-110 significantly reduced tumor growth in an animal colon cancer tumor model and was chosen for the PD1-Vaxx inhibitory vaccine.
Key findings:
- PD1-Vaxx outperformed the standard anti-mouse PD-1 antibody (mAb 29F.1A12) in an animal model of HER-2 expressing colon carcinoma;
- The combination of PD1-Vaxx with combo HER-2 peptide vaccine (B-Vaxx) showed enhanced inhibition of tumor growth in a HER-2-positive colon cancer model;
- Both the PD-1 and the combined vaccines were safe with no evidence of toxicity or autoimmunity.
“With additional study,” Kaumaya says, “we believe PD1-Vaxx will prove to be safer, more effective and have a lower incidence of resistance than checkpoint-blockade antibodies.”
This study was supported by grants from the National Institutes of Health (CA84356, CA13508, CA181115), and by Imugene Ltd. The safety of the vaccine was confirmed in pre-clinical animal studies at OSU and Charles River labs (Ashland, Ohio).
Vaccine Received IND Approval
In November 2020, the U.S. Food and Drug Administration (FDA) granted investigational new drug (IND) approval to Imugene for clinical testing of the investigational vaccine, known as PD1-Vaxx, an important milestone in the research collaboration between Ohio State and Imugene.
A first-in-human, phase1 clinical trial to test the vaccine is expected to open at the OSUCCC James in early 2021 for certain patients with non-small cell lung cancer. Additional U.S. sites may be added to trial at a later date.
“We are excited to begin testing of this vaccine in the United States to offer new hope to patients with lung and other cancers. Reaching this point where we can transition our findings from the lab to the clinic speaks to the perseverance and dedication of Imugene’s clinical and research team—including our research lab staff at Ohio State—to build on the clinical and commercial potential,” said Kaumaya.
Imugene’s Chief Executive Officer & Managing Director Leslie Chong, said “the multiple commercial, strategic and clinical benefits of our collaboration with the OSU secures our leadership position in the promising B-cell peptide cancer vaccine sector, and in particular PD-1 checkpoint inhibitors, where OSU’s pre-clinical work for a Phase I PD-1 clinical trial was pivotal to our FDA IND approval.”
Source: Read Full Article
