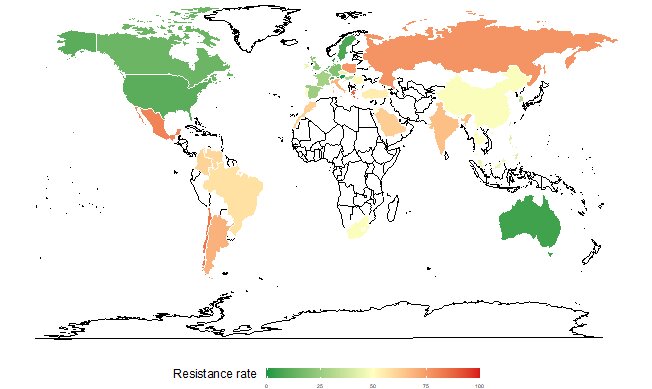
To understand the main determinants behind worldwide antibiotic resistance dynamics, scientists from the Institut Pasteur, Inserm, Université de Versailles Saint-Quentin-en-Yvelines and Université Paris-Saclay developed a statistical model based on a large-scale spatial-temporal analysis. Using the ATLAS antimicrobial resistance surveillance database, the model revealed significant differences in trends and associated factors depending on bacterial species and resistance to certain antibiotics.
For example, countries with high quality health systems were associated with low levels of antibiotic resistance among all the gram-negative bacteria investigated, while high temperatures were associated with high levels of antibiotic resistance in Enterobacteriaceae. Surprisingly, national antibiotic consumption levels were not correlated with resistance for the majority of the bacteria tested. The results suggest that antibiotic resistance control measures need to be adapted to the local context and to targeted bacteria-antibiotic combinations.
The results of the study were published in the journal The Lancet Planetary Health.
Antibiotic resistance (ABR) is currently one of the most urgent threats to global health. It is a natural phenomenon, but improper use of antibiotics is contributing to it by selecting resistance and complicating bacterial infection-control strategies. Worldwide surveillance of antibiotic resistance, especially under the aegis of WHO has been set up, and several databases have been created to record ABR worldwide, with the long-term aim of improving understanding of the causes to help tackle the phenomenon.
Antibiotic resistance varies considerably depending on the bacterial species, but a recent study estimated that in 2019, 1.27 million deaths worldwide were attributable globally to ABR and ABR was associated with 4.95 million deaths.
To identify the main factors associated with worldwide antibiotic resistance dynamics, a multidisciplinary research team at the Institut Pasteur developed a statistical model and analyzed antibiotic resistance data from the ATLAS database, which contains data collected since 2004 in more than 60 countries on every continent. The scientists analyzed the data by testing a large number of determinants to reveal the main factors of antibiotic resistance and understand how they relate to the dynamics observed worldwide.
“Research teams study how antibiotic resistance emerges in a bacterium in a Petri dish or in an individual, but we are currently lacking a population-level, global overview that can be used to investigate links between resistance and specific factors like national health system quality for different species of pathogenic bacteria. To understand the dynamics of antibiotic resistance, it needs to be studied at every level. That is what this study sets out to do,” explains Eve Rahbé, a Ph.D. research student in the Institut Pasteur’s Epidemiology and Modeling of Bacterial Escape to Antimicrobials Unit and first author of the study.
The first stage of the study was to select relevant factors that could influence antibiotic resistance dynamics. “Although some biological factors are known, it was also important for us to investigate hypotheses associated with socioeconomic and climate factors,” continues the scientist. A total of eleven independent factors were selected, including health system quality (based on the GHS index), antibiotic consumption and national wealth (GDP per capita), as well as data on travel and climate variables. Statistical models were then developed to study potential associations between the ATLAS data and the selected factors.
The analysis of global data for the period 2006-2019 initially revealed an increase in resistance to carbapenems for several species, although global trends were stable for other resistances. The study also demonstrated that the dynamics and factors associated with antibiotic resistance depend on bacteria-antibiotic combinations. Surprisingly, however, national antibiotic consumption was not significantly associated with resistance for the majority of bacteria tested (except for quinolone consumption for fluoroquinolone-resistant Escherichia coli and Pseudomonas aeruginosa and carbapenem consumption for carbapenem-resistant Acinetobacter baumannii).
Conversely, high health system quality was associated with low levels of antibiotic resistance in all the gram-negative bacteria tested. High temperatures were associated with high levels of antibiotic resistance, but only for Enterobacteriaceae (Escherichia coli and Klebsiella pneumoniae).
“This study reveals the wide range of factors leading to antibiotic resistance among different pathogenic bacteria at global level, and the need to adapt resistance control approaches to the local context (country, transmission context) and the specific bacteria-antibiotic combination,” concludes Philippe Glaser, Head of the Institut Pasteur’s Ecology and Evolution of Antibiotic Resistance Unit and co-last author of the study.
“Our statistical model can be applied to other databases, such as the WHO database. Improving understanding of resistance determinants, which differ from one country to the next and probably even vary among regions in the same country, is crucial and will be useful in adapting public health measures,” concludes Lulla Opatowski, a Professor at Université de Versailles Saint-Quentin-en-Yvelines, scientist in the Epidemiology and Modeling of Bacterial Escape to Antimicrobials Unit and co-last author of the study.
More information:
Eve Rahbe et al, Determinants of worldwide antibiotic resistance dynamics across drug-bacterium pairs: a multivariable spatial-temporal analysis using ATLAS, The Lancet Planetary Health (2023). DOI: 10.1016/S2542-5196(23)00127-4
Journal information:
The Lancet Planetary Health
Source: Read Full Article